Real-world evidence (RWE) in pharma: Use cases

February 25, 2026 12 min read
The health sector is producing more data than any other industry. The amount of data that the health industry generates globally can account for 30% or more of all the data produced worldwide (including electronic health records, registries, medical device data, claims data, and patient-reported outcomes), which means that pharmaceutical companies and pharmacy stakeholders are going to be impacted by this change in the way we create, interpret and use evidence. While clinical trials remain important and will continue to play a crucial role in evidence creation, they do not fully capture everything there is to know about a drug.
Real-world evidence (RWE) enables organizations to generate more detailed data on the safety and effectiveness of drugs, thanks to the increased size of the patient population and the longer duration of data collection. In 2026, generative AI (GenAI) is accelerating every stage of that process, from structuring messy unstructured data to automating study reports and detecting safety signals faster than any manual workflow could.
Key takeaways on RWE in pharma and clinical practice
- Real-world data (RWD) and RWE go beyond traditional clinical trials. They offer important practical insights into the real-world performance of innovative medical devices, particularly in larger and more diverse patient populations.
- Pharmaceutical companies can locate more effective clinical trial venues, relax eligibility requirements, and meet enrollment goals more quickly by utilizing observational health data. This entails finding the appropriate patients and making studies more feasible.
- Using EHRs to track and improve how doctors practice enhances both the quantity and utility of Real-World Data after a product enters the market. With this type of RWE, clinicians can identify essential trends earlier, receive more precise guidance, and deliver better care to their patients.
- GenAI is now embedded across the RWE lifecycle, helping pharma teams structure unstructured data, auto-generate study reports, and run safety signal detection at scale, compressing cycle times by an estimated 30–40%.
Real-world data sources and data quality considerations in pharma
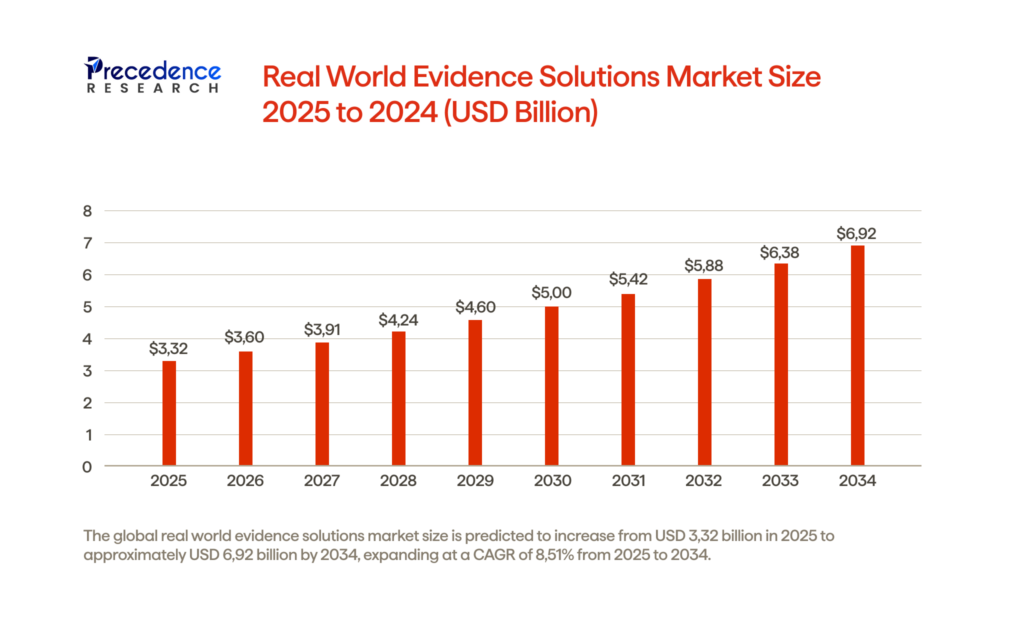
RWD has transitioned from being an ancillary input into pharmaceutical research to being a vital part of it. Collectively, the RWD/RWE industry is expected to be worth $3.32 billion in 2025, and estimates indicate that the market will grow to nearly $6.92 billion by 2034, representing an annual compound growth rate of 8.51%. This rapid growth reflects the deep integration of both RWD and RWE into the processes of drug development, regulatory approval, and subsequent optimization of patient outcomes. With an increasing reliance on real-world data insights, the industry is now addressing a fundamentally different question than before: not whether, but rather how to responsibly utilize this wealth of available information.
The most significant contributors of data in RWE are health records, insurance claims, disease registries, pharmacy dispensing data, wearable devices, and patient-reported outcomes. All of these different sources provide us with different lenses through which we can view the same thing. Claims provide longitudinal and large-scale data, registries provide disease-specific detail, while Electronic Health Records provide more detailed clinical information. All of these different sources of real-world evidence (RWE) enable RWE teams to observe and understand real-world treatment patterns, long-term safe and effective use, as well as variations in care that may not have been captured during clinical trials.
Broader data access comes with real challenges. Data collection across different health systems usually does not follow a set format (or standard). Furthermore, coding begins in other ways (e.g., “C” for “Codes”; “M” for “Missing”). Unless these issues are addressed in the early stages of data collection, the results will be skewed.
Governance is the foundation for a successful RWD program. It includes validating the source of data, establishing inclusion criteria, managing bias, and documenting provenance. Advanced analytics, AI-enhanced standardization, and interoperability frameworks facilitate alignment among various data sets, thereby enhancing data reliability.
Properly structured and high-quality RWD increases confidence in the generation of evidence. High-quality RWD also enables improved regulatory submissions, informed lifecycle management decisions, and ultimately results in better patient outcomes. The importance of RWE is determined less by the quantity of samples and more by the degree of trust, structural integrity, and scientific rigor associated with the data.
Use case 1: improving patient recruitment and clinical trial feasibility with RWD
One major barrier to undertaking clinical research is still patient recruitment. The failure of 80% of clinical trials to reach their enrollment goals will have a direct effect on your study’s completion schedule, budget, and the regulatory body’s perception of your results. According to Arnaub Chatterjee, Chief Product and Solutions Officer of Trinetx, “With over 80% of trials failing to meet enrollment targets, RWD offers a pivotal opportunity to enhance trial feasibility and site identification.”
Today’s enormous databases of real-world data, such as insurance claims, e-health records, illness registries, and shared patient data, have enabled researchers to track patient responses to treatments in real-time. They may keep an eye on how diseases develop, which helps identify the best candidates for clinical trial participation. Researchers can also quickly determine whether a patient meets the trial enrollment parameters and assess the viability of their proposals.
RWD enables more precise cohort definitions by filtering patients based on lab values, prior treatment failures, comorbidities, or progression patterns. Improved quality in both the enrollment process and downstream safety and efficacy assessments takes place due to the increased precision of the cohorts. This skill can reduce the number of mismatched populations by eliminating unexpected adverse events and unnecessary or costly protocol amendments.
TriNetX has enabled Chatterjee and his team to transform a complex protocol for a global infectious disease trial into actionable queries for the real world in under 24 hours. The result was 50 newly identified trial sites located throughout the United States, the United Kingdom, and Israel. The findings illustrate how effective, robust RWE has allowed for faster enrollment times, improved diversity, and trials that better reflect real patient populations.
GenAI at work in trial recruitment
GenAI takes feasibility analysis further by processing free-text clinical notes, radiology reports, and pathology records that structured queries miss entirely. Large language models can scan thousands of unstructured EHR entries, extract relevant patient characteristics, and flag eligible candidates in hours rather than weeks. Beyond identification, GenAI-powered tools can simulate trial scenarios using virtual patient cohorts — testing protocol assumptions before a single site is activated, reducing costly mid-trial amendments.
Use case 2: comparative effectiveness research to support regulatory approval
Real-world studies that measure the efficacy of a treatment as it is used in everyday clinical settings, employing comparative effectiveness research (CER), provide evidence to evaluate how well the therapy performs compared to other therapies. The type of information that CER generates provides regulators with insights that help fill gaps typically left by traditional clinical trials, particularly with respect to long-term outcomes, diverse populations, and safety after the product is used in a broader range of patients.
Well-constructed RWE programs enable pharmaceutical organisations to assess the impacts of their offerings through multiple avenues, including Electronic Health Records (EHRs), claims, and registries. By using these and other data sources to conduct head-to-head comparisons, they will be able to demonstrate value as part of label extension, reimbursement negotiations, and compliance with regulatory authority requirements for post-market commitments. Researchers will use these data to present a comprehensive and conclusive case regarding the real-world value of a drug beyond what was provided by clinical studies.
How RWE supports regulatory decision-making:
- Provides a comparison of outcomes for patient subgroups that are often underrepresented in clinical trials;
- Enables early identification of safety signals and rare adverse events;
- Allows for real-world measures of adherence and treatment switching;
- Allows for more than a single benefit-risk assessment across the longer term.
Together, CER and RWE provide regulators with practical, decision-ready insights grounded in real clinical use.
GenAI at work in comparative effectiveness
Systematic literature reviews, the backbone of any CER package, are traditionally labor-intensive. GenAI can automate abstract screening, data extraction, and evidence synthesis across hundreds of publications, cutting review timelines significantly. It also helps draft value dossiers and global reimbursement submissions by pulling together clinical, safety, and health economics evidence into consistent, regulator-ready narratives. According to TCS research presented at ISPOR 2024, GenAI-assisted regulatory operations can reduce approval cycle times by 30–40%.
Use case 3: post-approval safety monitoring using electronic health records
After a therapy is launched on the market, it becomes essential to understand how it behaves in the “real world” outside of the controlled conditions of the study. The collection of real-world evidence from electronic health records (EHRs) is a crucial component of ongoing safety monitoring, providing ongoing information about the effects of treatment on a wide range of patients over time.
The use of EHRs, as opposed to clinical trials, will provide a more comprehensive picture of a medication’s efficacy and safety. Since EHR data provide ongoing, routine information about treatment, treatment adherence, and the demographics of individuals, we can track larger patient populations who have been treated with the medication. By analyzing EHR data in this manner, we will have the ability to detect safety signals (such as unexpected adverse events); these signals can be detected earlier by collecting and analyzing the data at scale.
The process of EHR surveillance is also used to consider potential Adverse events before they occur. Using EHR analytics to identify potential Adverse Drug Events (ADEs) provides sponsors with early insight into possible problems, guiding the early detection of issues through studies or label changes based on the identification of ADEs. By using real-time and ongoing identification of ADEs, the connection between regulators and sponsors, as well as between sponsors and healthcare providers, is strengthened.
GenAI at work in safety surveillance
Traditional pharmacovigilance relies heavily on structured adverse event reports. GenAI expands the signal-detection surface by ingesting unstructured sources, patient forums, social media, clinical notes, and online health communities, and converting them into structured safety reports through natural language processing. Predictive models can then simulate how dose variations, drug interactions, or demographic shifts might affect adverse event rates, giving safety teams a proactive view of emerging risks rather than a reactive one.
Leverage cutting-edge tools to streamline and automate clinical trial processes. Get fast, reliable outcomes and expedite drug development.
Use case 4: optimizing clinical practice with real-world evidence
Once a therapy moves out of the clinical trial bubble and into the real world, things change fast. Suddenly, people use it in all sorts of ways—different ages, other health problems, some folks stick to the plan, some don’t, and every clinic does things a little differently. That’s where real-world evidence steps in. It demonstrates how these treatments effectively work for all types of patients, not just those who participate in trials. For pharma companies, this is gold. They can identify which patients actually benefit, when doctors need to adjust the dose or the order of treatments, and what success looks like when life isn’t so controlled.
Over time, these real-world insights do more than just sit in a report. They can drive label changes, update medical guidelines, and even inform the decision tools doctors use during appointments.
GenAI at work in clinical practice optimization
GenAI brings a new layer of intelligence to this process by summarizing patient histories at the point of care, giving clinicians a concise, AI-generated briefing before consultations rather than requiring them to read through hundreds of pages of records. Novo Nordisk’s Sophia chatbot is an early example: within six months of deployment, it handled 11,000 patient conversations and answered 27,000 questions outside business hours. As GenAI models become more clinically specialized, tools like Google’s MedPaLM are setting a benchmark for accuracy in medical question-answering — moving from research curiosity to clinical utility.
| Use case | Main goal | Typical data sources | What it enables | GenAI role |
|---|---|---|---|---|
| 1) Recruitment & feasibility | Faster, better-fit enrollment | EHRs, registries, claims | Site selection, eligibility tuning | Unstructured EHR parsing, virtual patient simulation |
| 2) Comparative effectiveness | Support approval decisions | Claims, EHRs, outcomes registries | Head-to-head insights, value evidence | Automated SLR, dossier drafting |
| 3) Post-approval safety | Detect risks earlier | EHRs, claims, pharmacovigilance | Signal detection, risk mitigation | NLP-based AE extraction, predictive modeling |
| 4) Clinical practice optimization | Improve real-world outcomes | EHRs, registries, patient-reported data | Pathway refinement, subgroup guidance | Patient history summarization, AI chatbots |
FAQ
Conclusion: How real-world evidence is reshaping pharma decision-making
Real-world evidence is turning into pharma’s second set of eyes after clinical trials, and GenAI is sharpening the lens considerably. It checks how therapies hold up in the real world, spots rare safety issues earlier, and surfaces the patient-level insights that trials rarely capture.
By 2026, the competitive edge will belong to organizations that do not just collect more health data but build the AI-augmented infrastructure to turn it into reliable, repeatable, decision-ready evidence, clean data pipelines, GenAI-assisted study design, and insights that physicians and regulators genuinely trust. Avenga helps life sciences organizations build exactly that.
Are you interested in finding out more about RWD and RWE in the pharmaceutical sector?
Contact Avenga, your trusted technology partner.